Government Recommends Registration of Breakthrough HIV Prevention Drug Lenacapavir
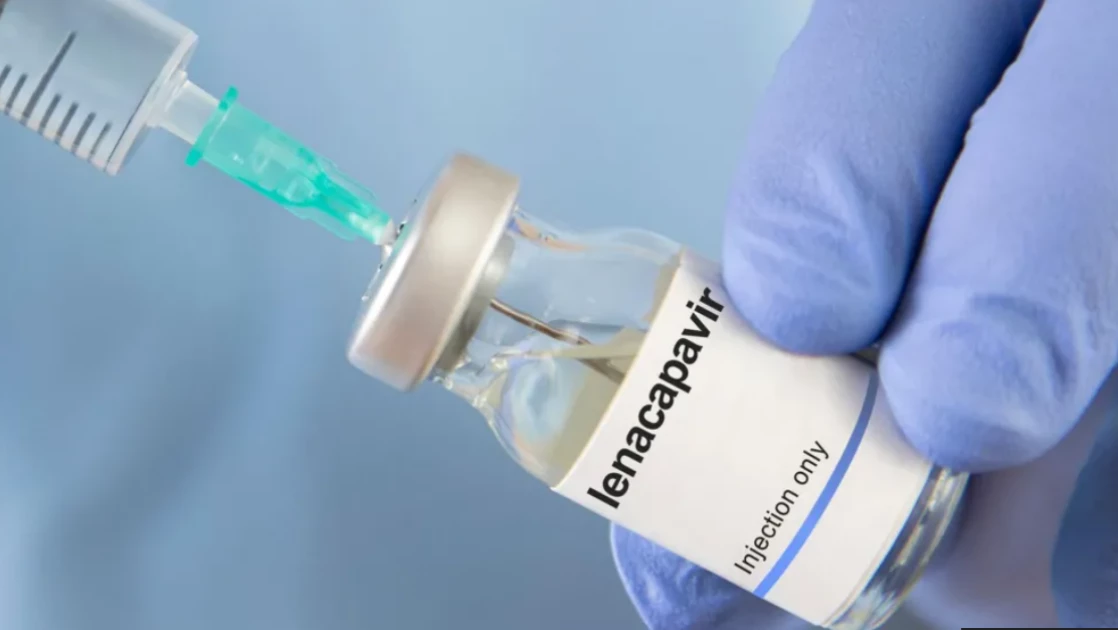
The Ministry of Health has recommended the registration of Lenacapavir, a groundbreaking long-acting HIV prevention medication, as part of Kenya’s intensified efforts to curb new infections.
In a statement issued on January 9, 2026, the Ministry, through the Pharmacy and Poisons Board, announced its recommendation for the registration of Lenacapavir 300 mg tablets and Lenacapavir 464 mg solution for injection. This long-acting antiretroviral is intended for use as HIV pre-exposure prophylaxis (PrEP).
The decision was reached after a thorough scientific evaluation of the drug’s quality, safety, and effectiveness, in full compliance with Kenyan regulations and international standards.
“The recommendation follows a comprehensive scientific assessment of the quality, safety, and efficacy of Lenacapavir, conducted in line with Kenyan law and applicable international regulatory standards,” stated Health Cabinet Secretary Aden Duale.
CS Duale explained that Lenacapavir works by interrupting critical stages in the HIV life cycle, thereby preventing infection. Unlike conventional daily oral PrEP tablets, this innovative injectable formulation is administered only twice a year, making it an attractive option for individuals who find daily pill-taking difficult.
“Its long-acting formulation allows it to be administered only twice a year, offering an important alternative to daily oral HIV prevention medicines… particularly beneficial for individuals who face challenges with taking pills every day,” noted CS Duale.
With this move, Kenya positions itself among the first African countries to recommend Lenacapavir for registration. The government views the decision as consistent with global best practices in HIV prevention



